Utilize the power of infectious disease models to inform your analyses and help you better understand the potential impact of your novel healthcare intervention.
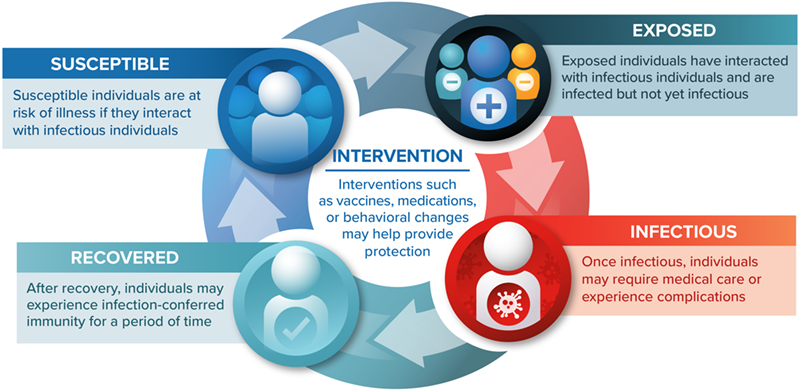
You rely on realistic disease models to understand and communicate the value of your healthcare products to stakeholders. Static health economic models are a useful tool, but they can only assess the direct impact of an intervention. When developing medicines, vaccines, or diagnostics for communicable diseases, you need to consider additional factors such as human behavior and herd immunity to effectively demonstrate the full impact of your product. Infectious disease models can capture indirect effects and population dynamics to help you better understand transmission of a contagious disease and enable you to present your product to its full potential.
Understand how your intervention affects disease transmission, health outcomes, resource use, and costs.