There are daily medications available to treat symptoms of Parkinson's disease (PD) - such as shakiness, stiffness, and difficulty moving. As PD progresses though, most patients experience periods during the day of "OFF" episodes (worsening of symptoms), even when taking one of these medications.
The US Food and Drug Administration has approved three on-demand treatment therapies to relieve these episodes. It was not understood, however, which aspects of the treatments patients value most. Researchers with RTI Health Solutions conducted a patient-preference study using a discrete-choice experiment survey to elicit this information.
The primary objectives were to quantify patient preferences for PD OFF‑episode treatment features and estimate the relative importance of features to patients. The survey presented patients with a series of hypothetical treatments to choose between. The treatments were based on different combinations of values from four treatment features of on-demand therapies.
- Method of administration along with the level of adverse effects
- Time to full "ON" (the time it would take to relieve symptoms)
- How long symptom relief would last
- Cost
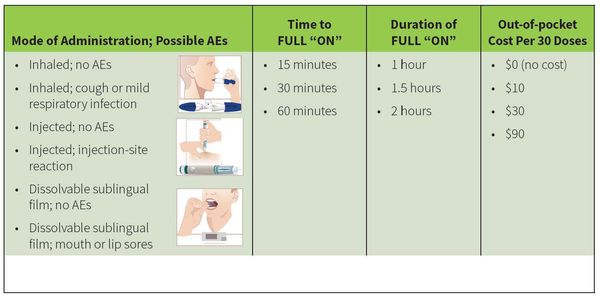
Overall, respondents placed the most importance on avoidance of high out-of-pocket costs and mode of administration. On average, the respondents were more likely to select the sublingual film treatment compared with the injection or inhalation powder treatments. They were also willing to pay more for the sublingual film and treatment that decreased the time it took to relieve symptoms.
Joshua Coulter, Research Economist at RTI Health Solutions, shared, "The results of this study can help future research focus on therapies with treatment attributes that patients with Parkinson's disease deem most important."
Thach A, Sutphin J, Coulter J, Mansfield C. A discrete-choice experiment evaluating preferences for on-demand treatments for patients with Parkinson's Disease and "OFF" episodes. Poster presented at the MDS Virtual Congress 2020; September 11, 2020. [abstract] Mov Disord. 2020 Sep; 35(S1):S427. doi: 10.1002/mds.28268.
